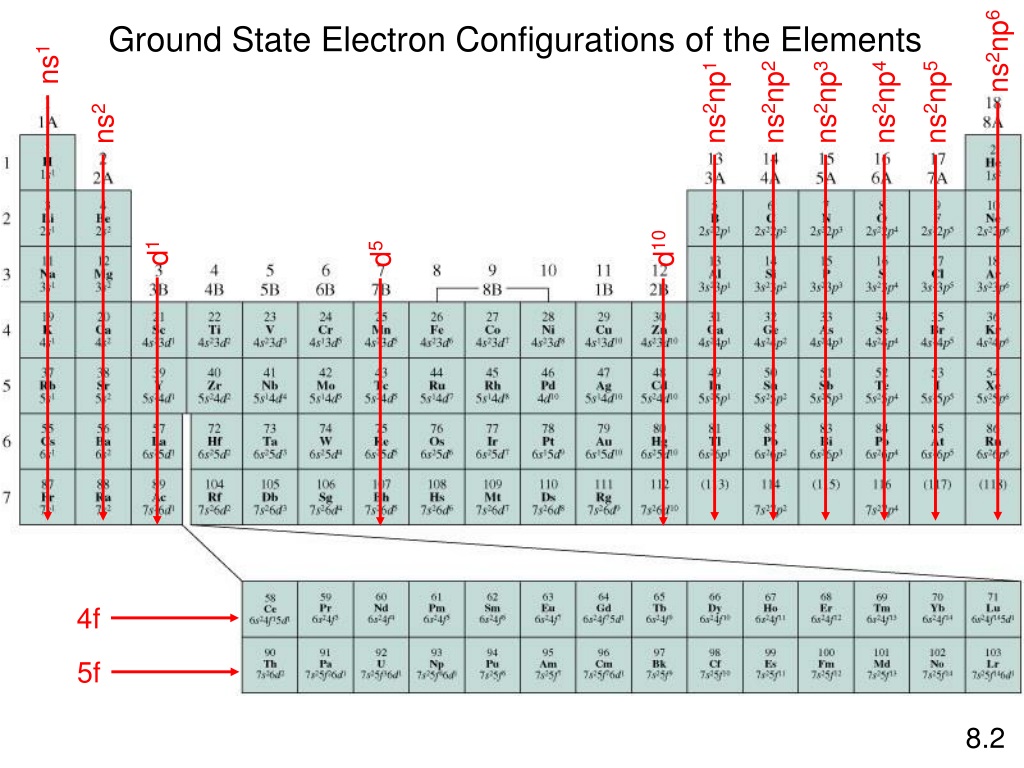
nuclear charge increases by one unit in each succeeding element but the corresponding addition of electron takes place in the same enthalpy shell. Reason: As we move from left to right in a particular period, the atomic number i.e. Department of Chemistry Jijamata College of Science and Arts, Bhende bk. Note that it is not possible to specify one element. So, if you were to find what elements have 5 electrons in the p orbitals, youd discover that any element in Group 7A (Florine, Chlorine, Iodine, etc.) satisfies this requirement. Valence-shell configuration: ns2 np3 Exhibits varied chemical properties. The presence of eight electrons in the valence shell of an atom imparts stability to that particular atom. This means that as we move across the 6 elements of the p bloc, we add one electron to the electron configuration. Valence electrons or outer-shell electrons are indicative of an atom’s reactivity. As we move from left to right in a period, atomic radii go on decreasing. The electronic configuration of nitrogen is \(2,\,8,\,7.\) Chlorine has \(7\) electrons present in its valence shell. Hence atomic radii of elements increase with the increase in atomic number as we move from top to bottom down a group. The effect of the progressive addition of a new shell outweighs the effect of increased nuclear charge. (ii) As we move down a group, a new enthalpy shell is added at each succeeding element though the number of electrons in the valence shell the remain the same. (i) Down a group, the nuclear charge goes on increasing. Ans: This is an outermost electronic configuration of an atom. As we move down a group in the periodic table, the atomic radii go on increasing. (ends in.) Valence e- e- Dot 1 ns1 2 ns2 13 ns2 np1 3 14 ns2 np2 4 15 ns2 np3 5 16 ns2 np4 6 17 ns2 np5 7 18 ns2 np6 8 Teacher: Fill in e-dot column with students in their notes.Variation of atomic radii in a group. Group 1 has 1 valence electron, Group 2 has 2 VE.….etc. The group number of a main group element tells us the number of valence electrons. What group in the periodic table has the valence shell configuration ns2np3 Group number 15 of Nitrogen family has ns2np3 valence shell electronic configuration. When done, count the electrons in the outer shell. Remember: The number of protons is equal to the atomic number. Since the element is not carrying a charge, there are equal numbers of protons and electrons. Valence electrons are the only electrons used in chemical bonds. It is the number of valence electrons that will determine the chemical properties of the element. Valence electrons are the electrons in the highest occupied energy level of an atom. Atoms of which group in the periodic table have a valence shell electron configuration of ns2 np3OpenStax is a registered trademark, which was not involved. Hibridisasi dari NH3 sp3 Electron kulit valensi atom 7N ns2 np3 Hibridisasi sp3 Atom N harus menyediakan 3 orbital dengan electron yang belum berpasangan, yang akan bertumpang tindih (berpasangan dengan elektron dari 3 atom H. Second, determine the number of valence electrons Draw the appropriate number of dots around the symbol making sure to put one dot on each side before putting a second dot. Lewis Dot Structures Used to show an element and the number of valence electrons.Ģ Writing Lewis Dots First write the chemical symbol M3 ions) : As the valence shell configuration of group 15 elements is ns2 np3. Presentation on theme: "Used to show an element and the number of valence electrons."- Presentation transcript:ġ Used to show an element and the number of valence electrons. p-orbitals in the valence shell of these elements are half filled.
